Show the TLC when Naphthalene (a neutral unknown compound) is compared to the correct (or matching) standard and what the TLC would look like if Naphthalene (neutral unknown compound) was compared to

NICS(1) values for naphthalene (1c) and azulene (2c), together with a... | Download Scientific Diagram
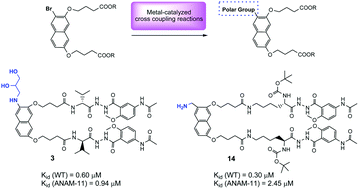
Introduction of polar groups on the naphthalene scaffold of molecular tongs inhibiting wild-type and mutated HIV-1 protease dimerization - MedChemComm (RSC Publishing)
SOLVED: what is the arrengement of the compounds of Perylene dianhydride, Naphthalene and Salicylic acid in order of increasing polarity by looking the structures and Rf values of them. Perylene dianhydride, Naphthalene
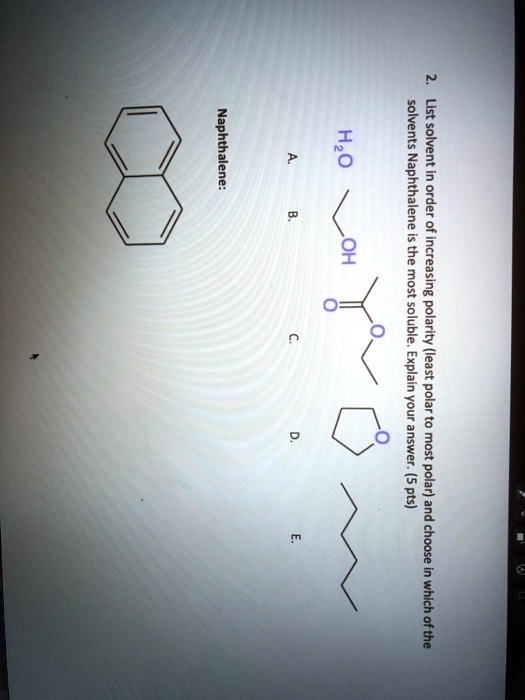
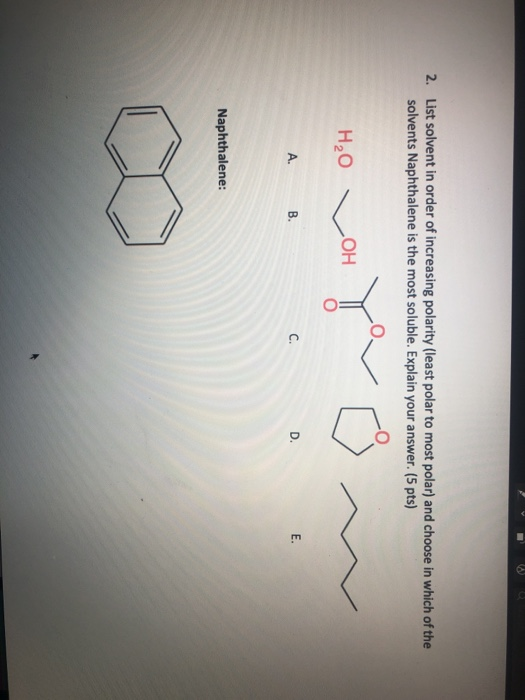
SOLVED: Naphthalene: Hz0 List solvent In OH solvents Naphthalene is the most soluble order of Increasing polarity (lepitinooura (least answer; most polar) pts) and choose which ofthe

Naphthalene and pDCB are non-polar compounds that are more weakly bound... | Download Scientific Diagram
Benzene derivatives and naphthalene solubilized in micelles. Polarity of microenvironments, location and distribution in micelles, and correlation with surface activity in hydrocarbon-water systems | The Journal of Physical Chemistry

Valli took some common salt, naphthalene balls, camphor, baking soda and washing soda. She attempted to dissolve these substances either in water or in acetone. Complete the table with the expected results.

What structural feature(s) do toluene and naphthalene have in common? State a reason for the observed solubility patterns of toluene and naphthalene in water and hexane. | Homework.Study.com


![3-Hydroxy-4-[(4-sulphonaphthyl)azo]naphthalene-2,7-disulphonic acid | C20H14N2O10S3 - PubChem 3-Hydroxy-4-[(4-sulphonaphthyl)azo]naphthalene-2,7-disulphonic acid | C20H14N2O10S3 - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=13507&t=l)