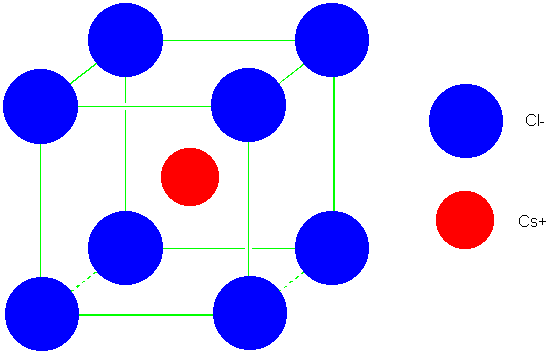
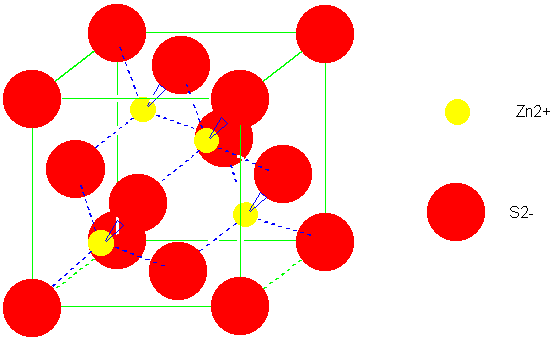
Write the first five terms of the Madelung constant for a two-dimensional lattice of alternating positive and negative ions. | Homework.Study.com

Generating the crystal structure samples for the Madelung constant test | Download Scientific Diagram

21. LECT21 - Madelung constant and the Born - Madelung constant and the Born-Lande eqn Lattice - Studocu
Show that the Madelung constant for a one-dimensional array of ions of alternating sign with - Sarthaks eConnect | Largest Online Education Community

Calculate the lattice energy of sodium chloride crystal from the following data: Born exponent, n=8, Madelung constant for NaCl=1.748, Ionic radius of Na^(+)=0.95 Å, Ionic radius of Cl^(-)=1.81Å.

solid state physics - Madelung constant for a two dimensional square crystal lattice - Physics Stack Exchange