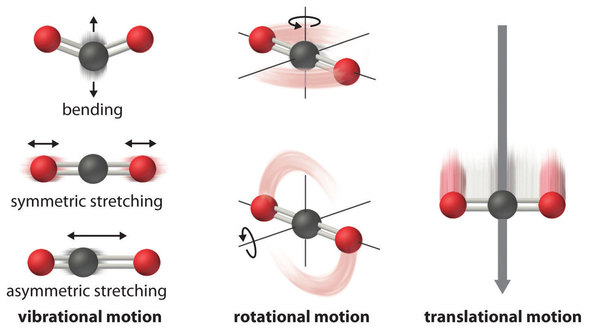
SOLVED: For each of the following, determine if the entropy change is negative O positive: Cleaning YOUI room HO) 7 HzOa) 4 S < 0 4s>0 04<0 4S>0 Heating solid NaClaq) `

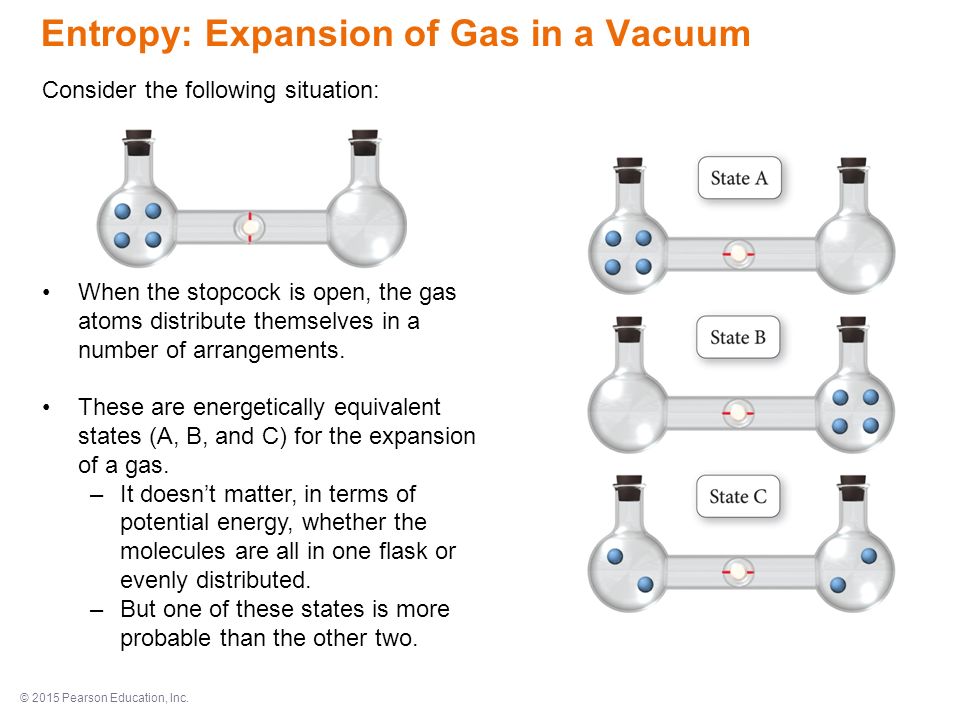
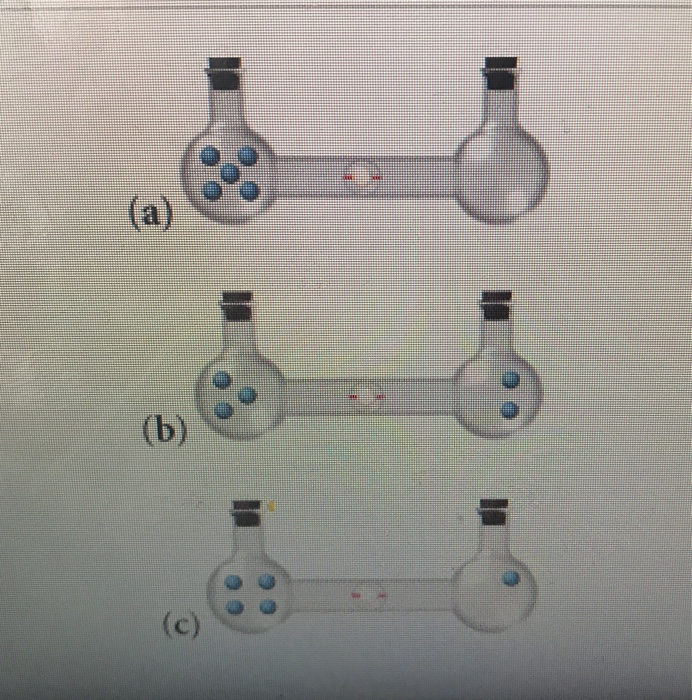
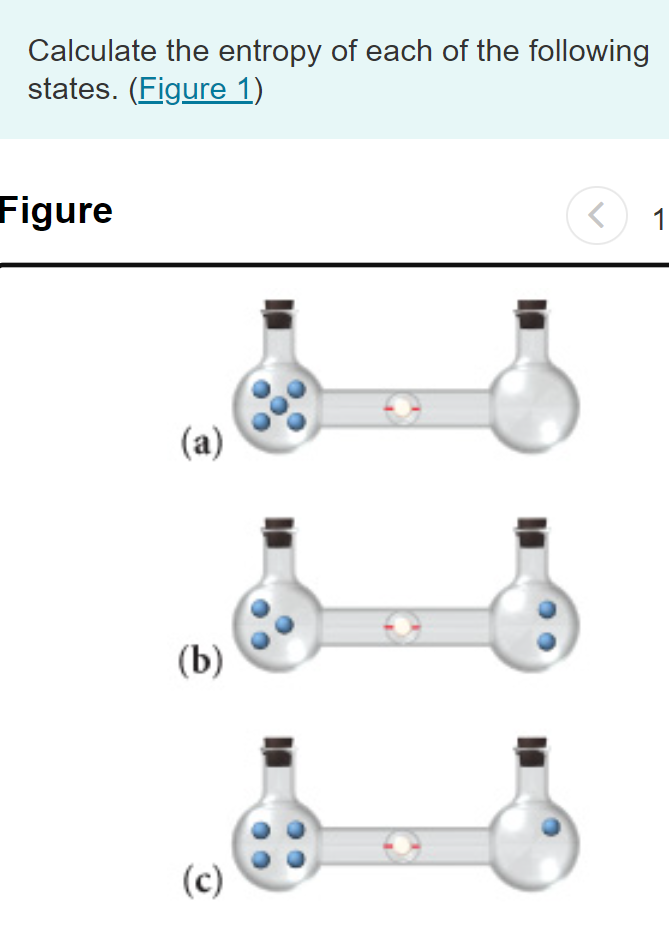
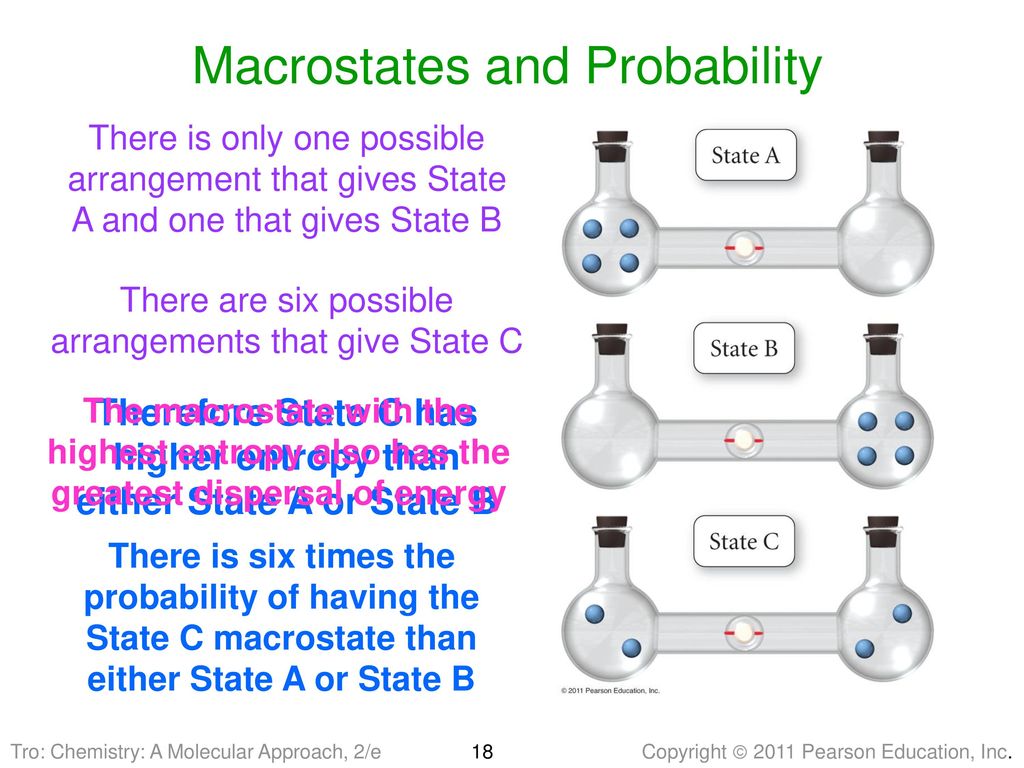
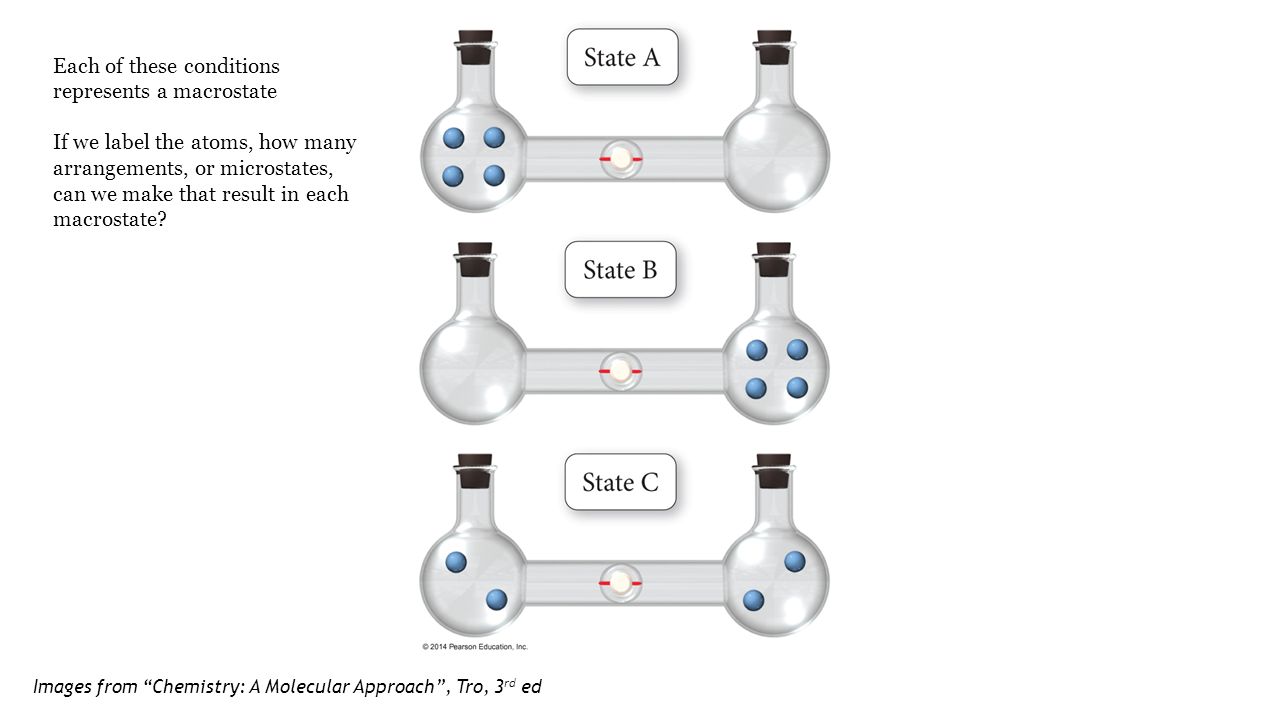
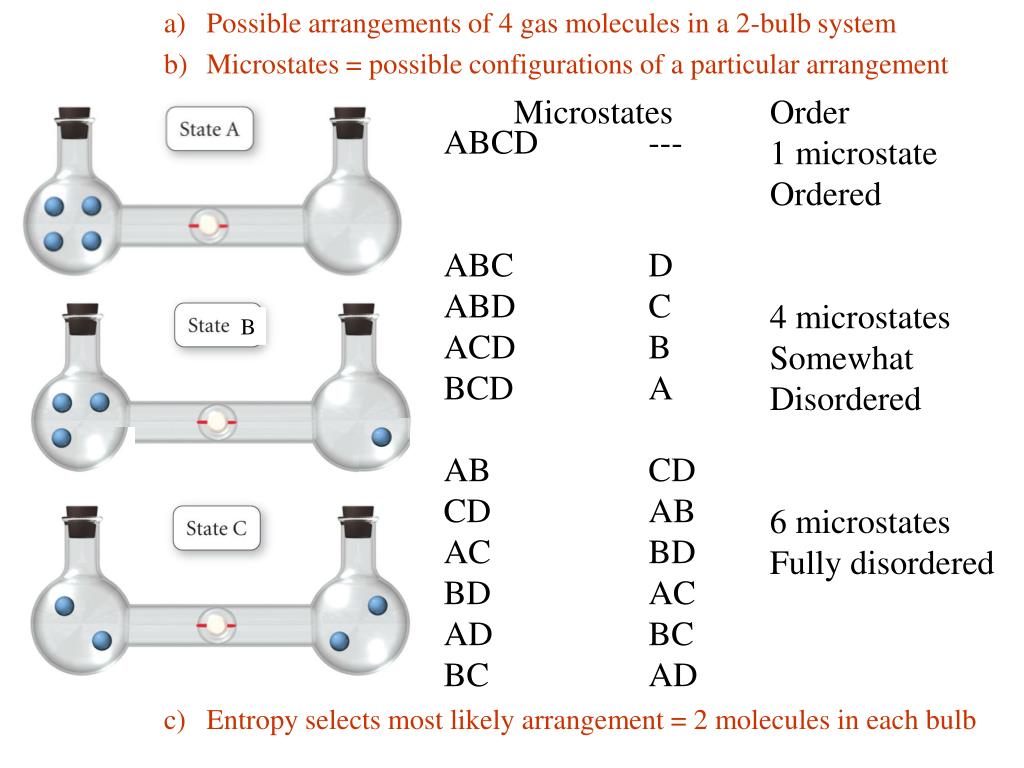
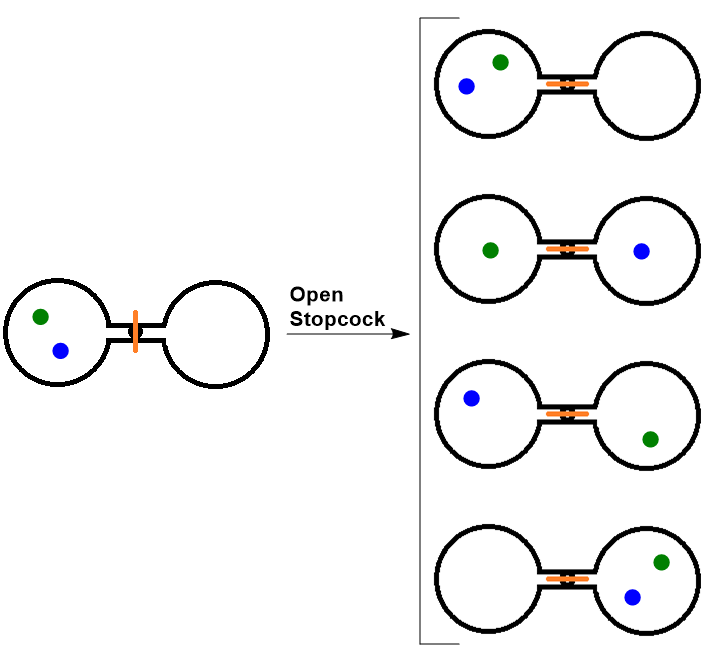
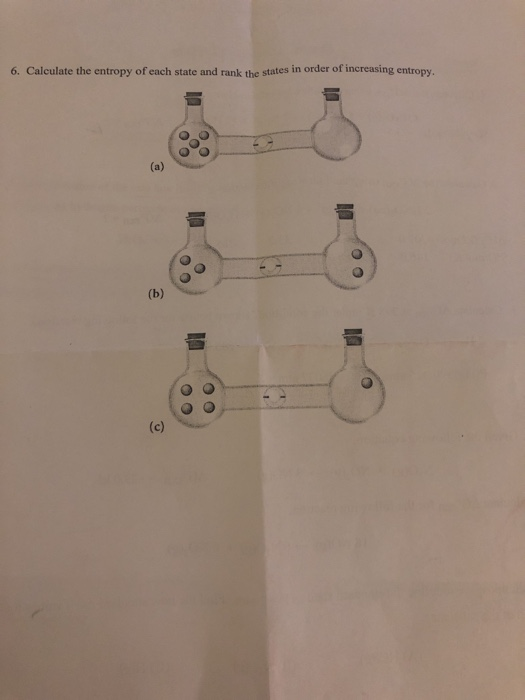
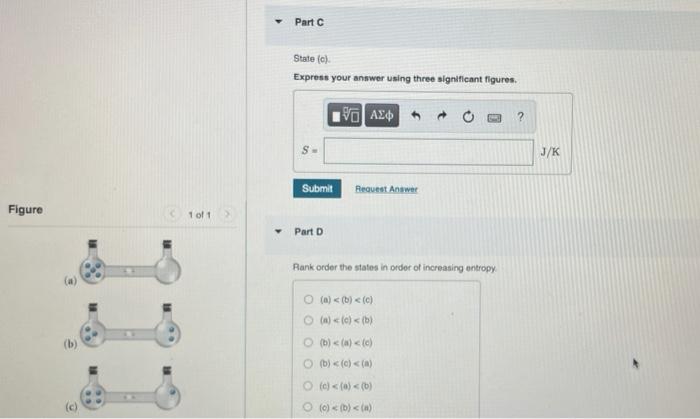
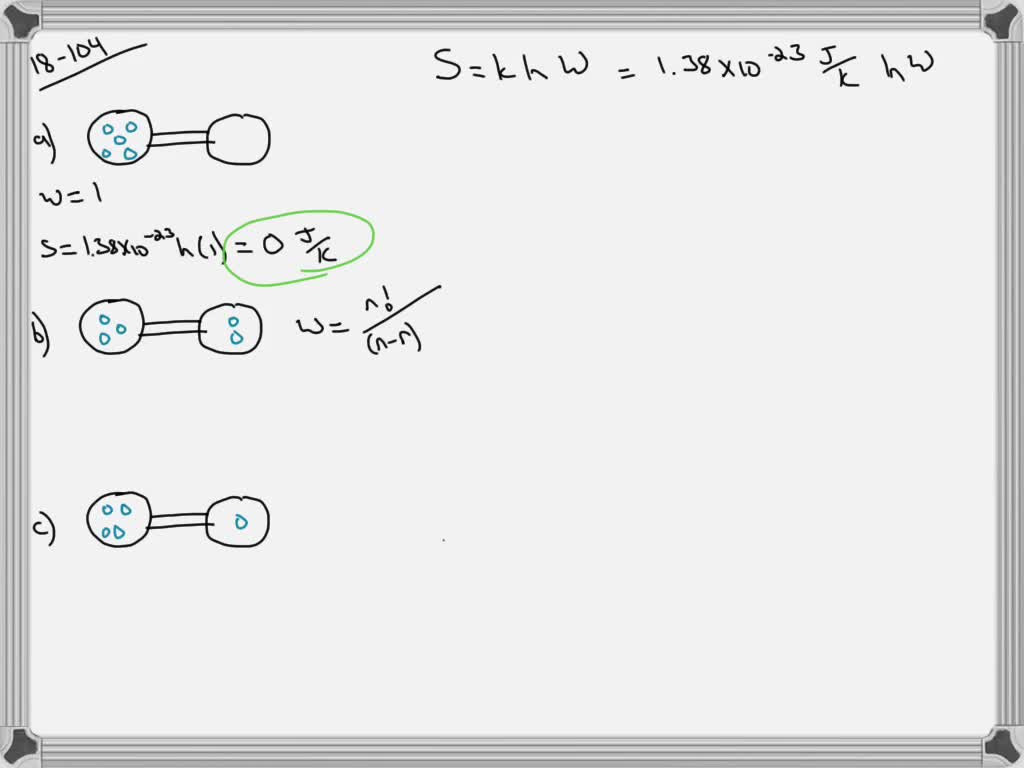
SOLVED:Calculate the entropy of each state and rank the states in order of increasing entropy. a. b. c.

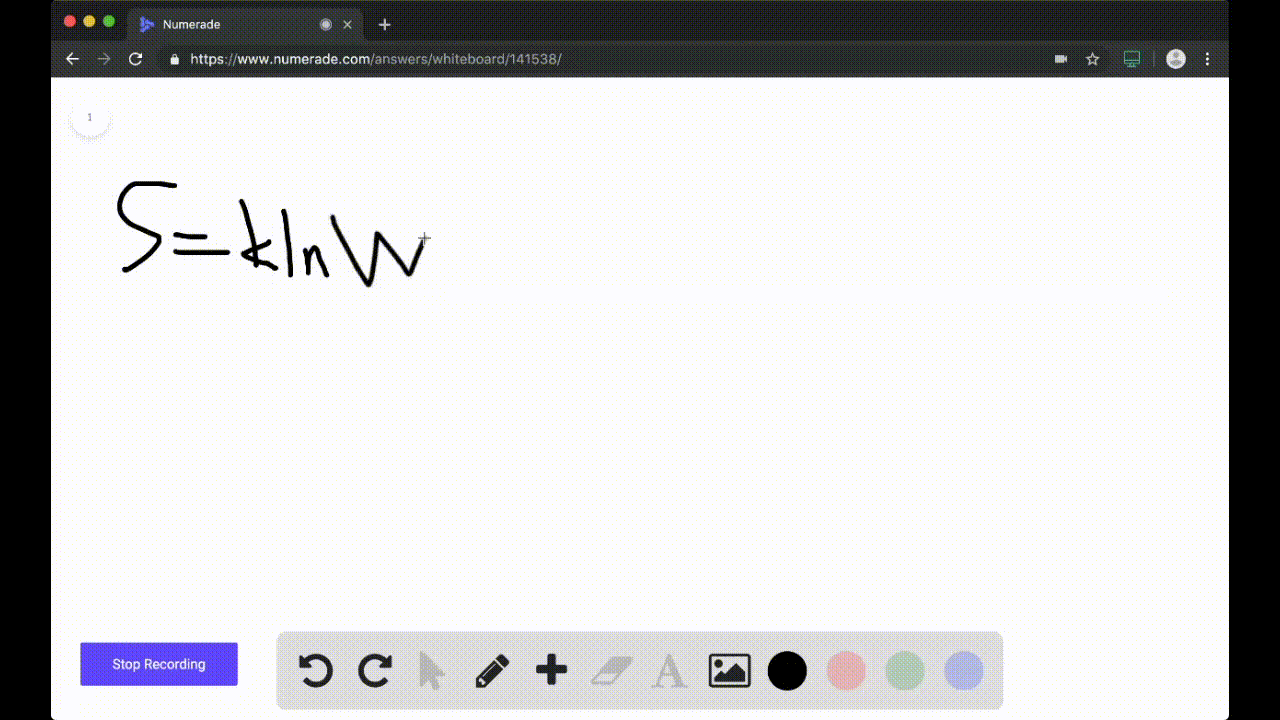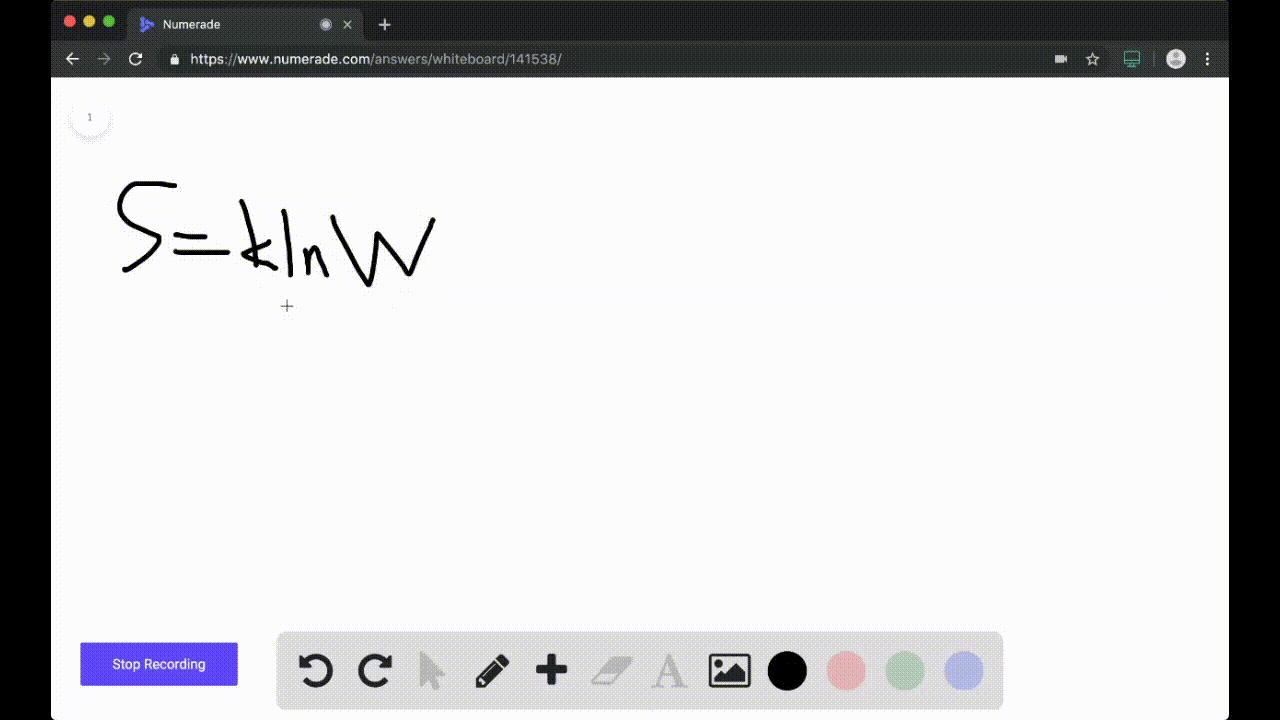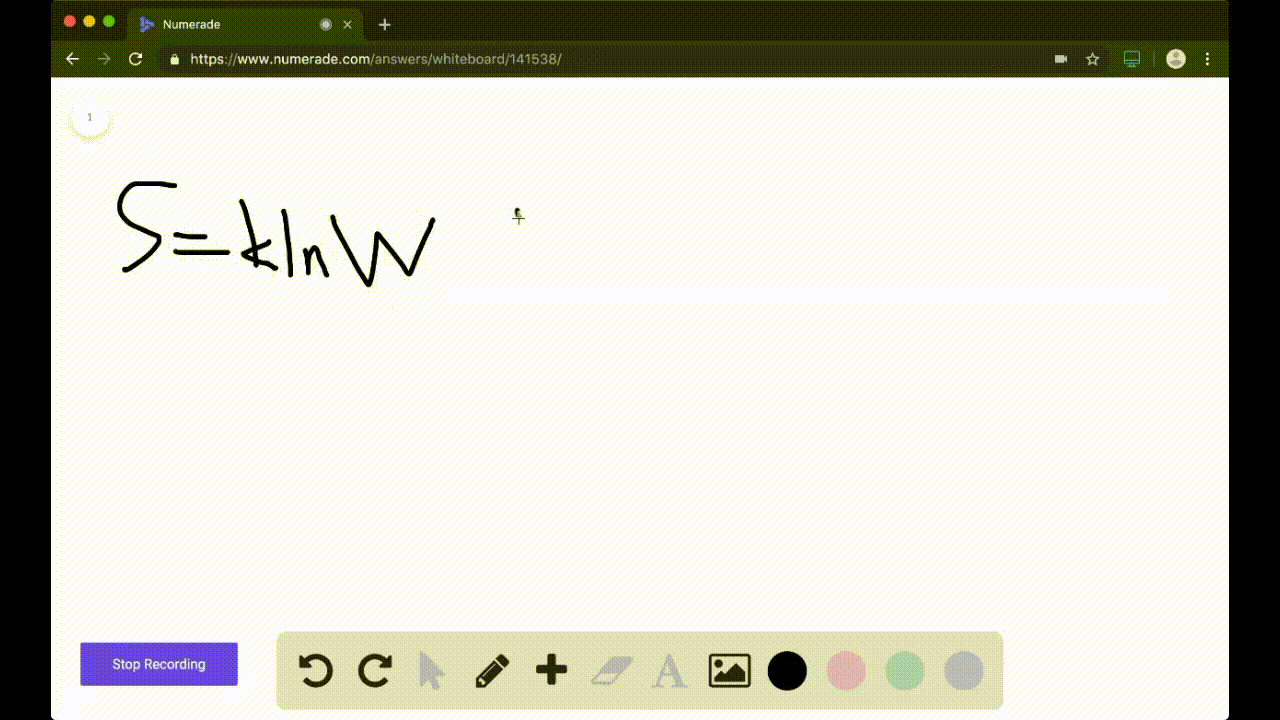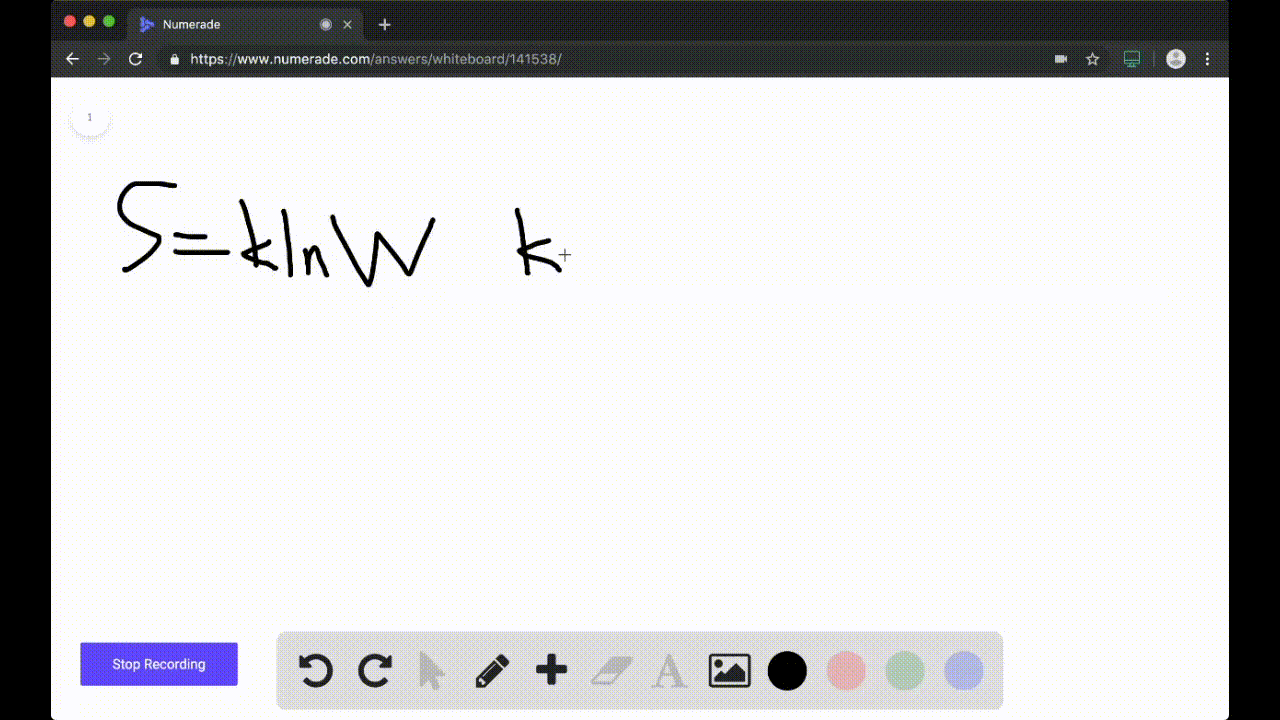
Calculate the entropy change for the following reaction, H2(g) + Cl2(g)⟶ 2HCl(g) at 298 K . Given that, S^ H2 = 131 J K^-1 mol^-1 , S^ Cl2 = 223 J K^-1 mol^-1 and S^ HCl = 187 J K^-1 mol^-1 .