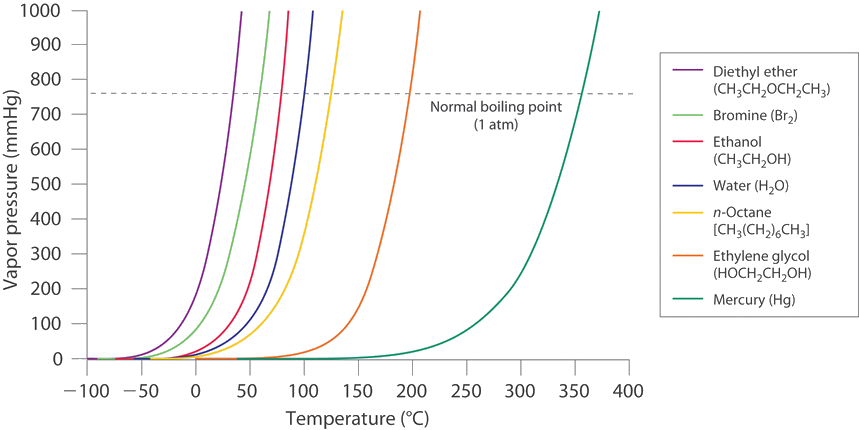
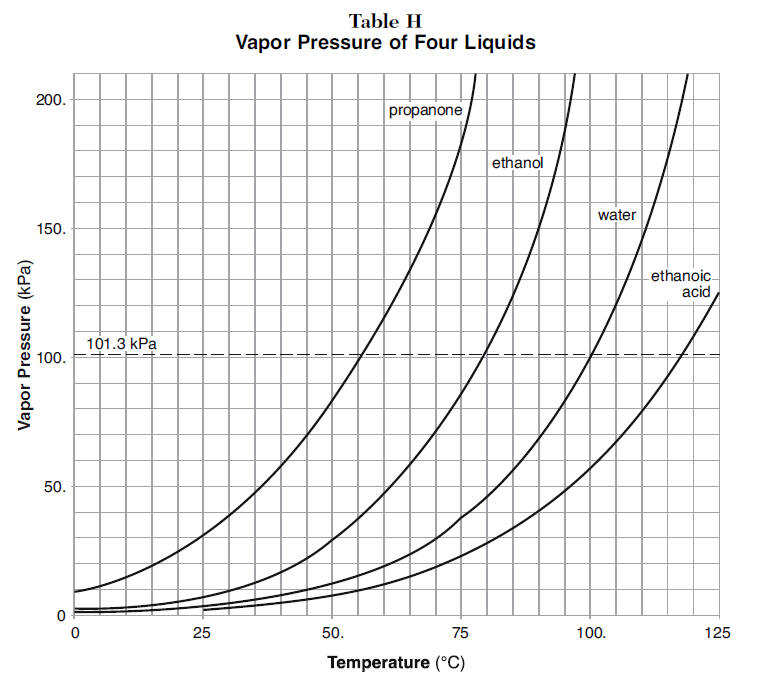
The vapour pressure of a pure liquid at 25 is 100 mm Hg . Calculate the relative lowering of vapour pressure if the mole fraction of solvent in solution is 0.8.

SOLVED:Suppose the vapor pressure of a substance is measured at two different temperatures. (a) By using the ClausiusClapeyron equation, Equation 11.1, derive the following relationship between the vapor pressures, P1 and P2,
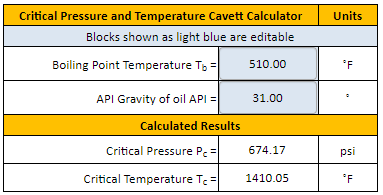
How does one calculate the boiling point of water at different pressures without a steam table? - Quora